
-
Products
Congenital Heart Disease Products
- MemoCarna® Atrial Septal Defect (ASD) Occluder
- MemoPart™ Atrial Septal Defect (ASD) Occluder
- MemoPart™ Ventricular Septal Defect (VSD) Occluder
- MemoPart™ Patent Ductus Arteriosus (PDA) Occluder
- MemoPart™ Patent Foramen Ovale (PFO) Occluder
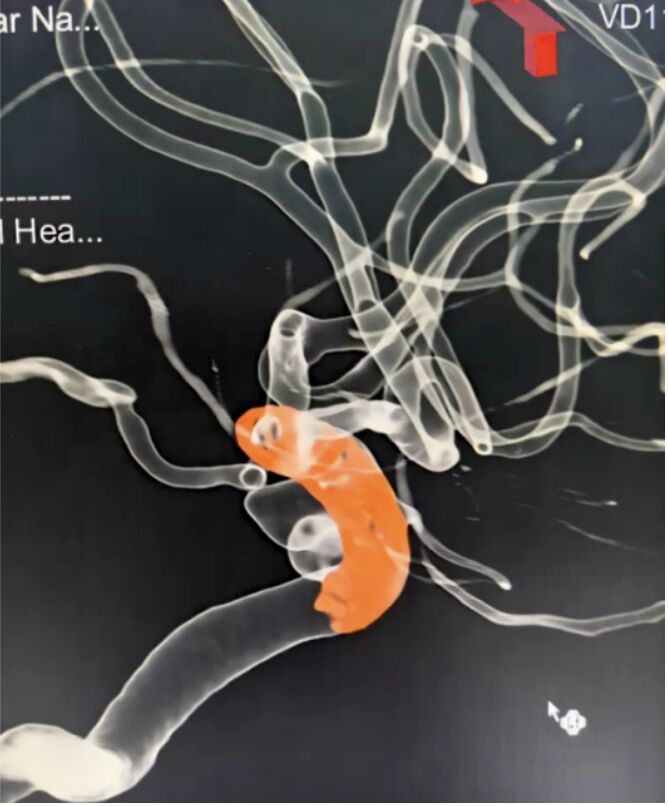
- MemoLefort®Left Atrial Appendage (LAA) Occluders System
- MemoPart™ Plug
- MemoPart™ Snare
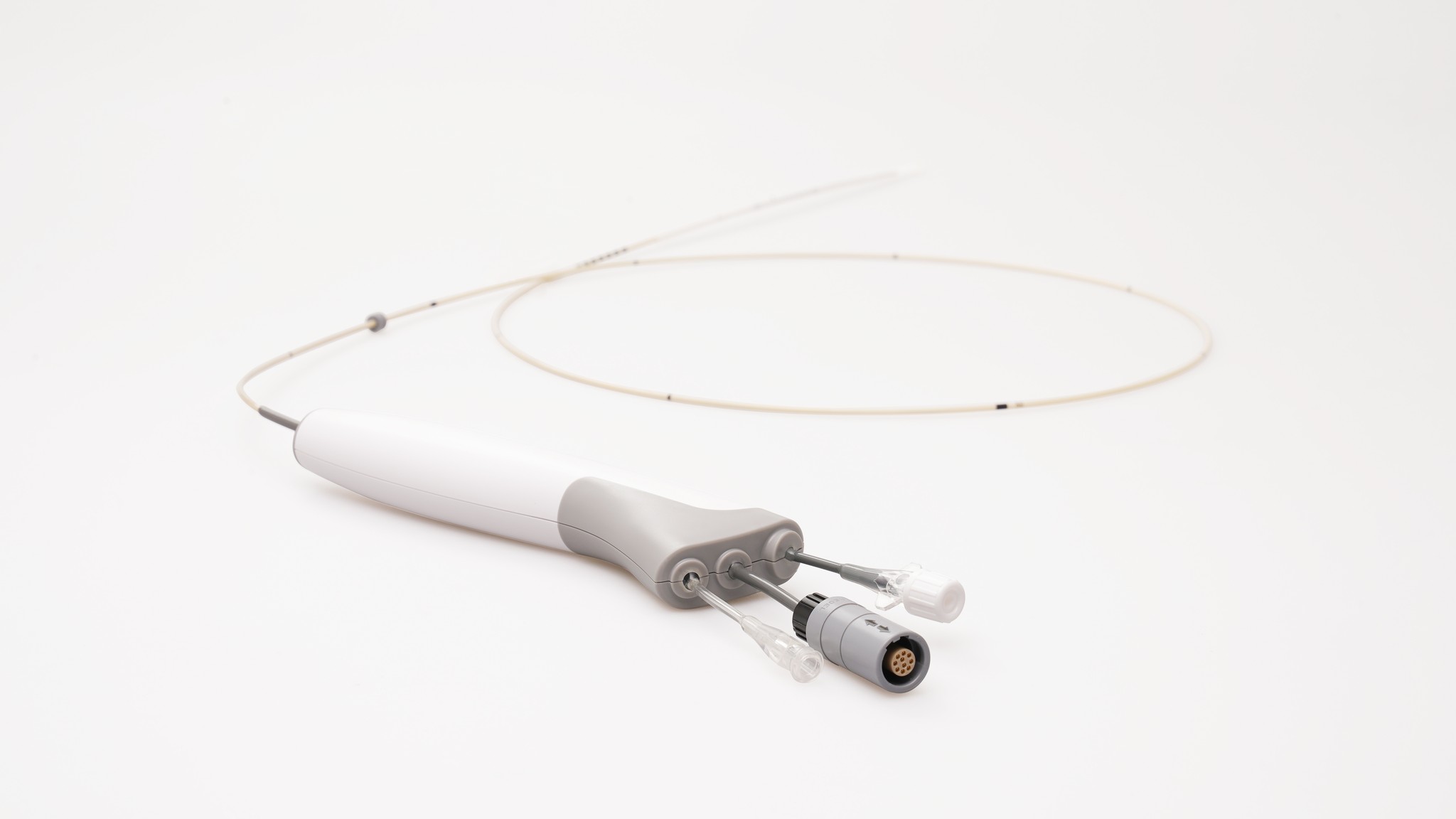
- MemoPart™ Occluder Delivery System
- Atrial Septal Defect Occluders
- Ventricular Septal Defect Occluders
- Patent Ductus Arteriosus Occluders
- Patent Foramen Ovale Occluders
- Occluder Delivery System
- Snare
- Plug
- MemoPart Atrial Septal Defect Occluder (ll-Hubless)
- MemoPart® Ventricular Septal Defect Occluder (II-Hubless)
- MemoPart® Patent Ductus Arteriosus Occluder (II-Hubless)
- MemoPart® Patent Foramen Ovale Occluder (II-Hubless)
- MemoPart® Plug (II-Hubless)
Critical Care Products- Safecath™ Disposable Central Venous Catheter Kit
- Safecath Plus Antimicrobial Central Venous Catheter Kit
- Disposable Hemodialysis Catheter Kit
- Disposable Blood Pressure Transducer
- Pressure-Resistant Peripherally Inserted Central Catheter Kit
- Arterial Blood Sampling Kits
- Needle-free Connectors
- CVC Accessories
- Disposable Precise Filtering Infusion Set with Needle
- Sterile Safety IV Catheter--ZModel
- Z Model Pen-like
- Pen Injector
- Pen Needles
- Disposable Postoperative Pain Relief System
- Disposable Non-phthalic Infusion Set with Needle
- Disposable Precision Filter Light-Proof Infusion Set With Needle
- Disposable Double-Layer Infusion Set
- Disposable Burette Infusion Set With Needle
- Anesthesia Product Catalog
SARS-CoV-2 Test Kit- SARS-CoV-2 Antigen Rapid Test Kit (Colloidal Gold Immunochromatography)
- SARS-CoV-2 Antibody Test Kit (Colloidal Gold Immunochromatography)
- Lepu SARS-CoV-2 Antigen Rapid Test Kits for Self-testing
- SARS-CoV-2 Antigen Rapid Test Cassette (Colloidal Gold Immunochromatography)
- 3-in-1 (Covid-19, Flu A, Flu B) Antigen Detection Kit
2019-nCoV Neutralization Antibody Test Kits- 2019-nCoV Neutralization Antibody Test Kit (Fluorescence Immunochromatography)
- 2019-nCoV Neutralization Antibody Test kit (Colloidal Gold Immunochromatography)
- 2019-nCoV Neutralization Antibody Test Kit (ELISA)
- 2019-nCoV Neutralization Antibody 2nd Gen Rapid Test Kit (Colloidal Gold Immunochromatography)
- Featured Solutions
- About Us
- Investors
-
e-IFU
- Contact Us

(1).jpg)



 Email Us:
Email Us: