Lepu Medical Develops Fluorometric Quantitative Reagents for Detecting Monkeypox Virus
-
2022-08-11
-
LEPU
According to the WHO news announcement, the monkeypox epidemic has found about 26,000 confirmed cases of monkeypox in more than 88 countries and regions, constituting the highest level of alert of "public health event of international concern (PHEIC)"! This has raised concerns about the spread of the virus. According to the WHO, Europe is currently a high-risk area for monkeypox, and the rest of the world is at a medium-risk level.
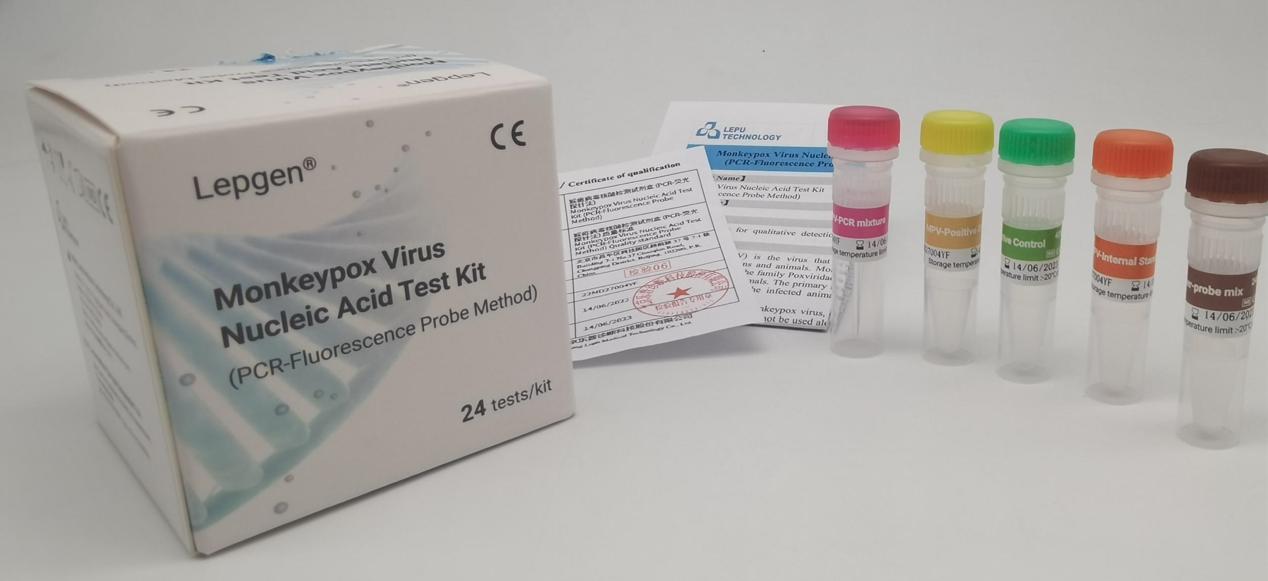
In response to the global outbreak of monkeypox, Lepu Medical developed a nucleic acid test kit (PCR-Fluorescence Probe Method) for the monkeypox virus and obtained the EU CE certification. This reagent provides a solution to help laboratories in various countries efficiently carry out monkeypox nucleic acid tests and rapid diagnosis.
Lepu Medical's monkeypox virus detection kit has the characteristics of high detection sensitivity, easy operability and strong stability. The kit can be quickly applied on multiple platforms such as Lepgen-96, Roche LightCycler 480 series, Bio-Rad CFX96, Shanghai Hongshi SLAN, and ABI 7500. At present, Lepu Medical has received orders and inquiries from many overseas countries and, at the same time, actively promotes the registration of this product in other overseas countries.