- Coronary Stents Coronary Balloons Catheters Guide Wires Introducer Sheaths Accessories Peripheral Interventional Products
- Immunoassay Molecular Diagnostics Hematology Diagnostics POCT Lab Solution
- Structural Heart Diseases Products
- Vascular Access Blood Pressure Transducer Arterial Blood Sampler CVC Accessories IV Catheter Needle-free Connectors Insulin Injection Pain Relief
- Hemodialysis consumables Hemoperfusion Cartridge Dialysis machine
- ECG Patient Monitor Oximeter
- Cardiovasculars Oncology Anti-infectives Central Nervous System Others
- Surgical Staplers Endoscopy Endoscopic Ultrasound System
- Trauma Implant Spinal Implant Artificial Joint Sports Medicine
- Pacemakers and Leads Equipments
- ELISA CLIA FIA
- Nucleic Acid Extraction Nucleic Acid Extraction & PCR Kits Real-Time PCR
- Thrombelastography Analyzer Blood Grouping Platelet Aggregation
- POCT - FIA Colloidal Gold Blood Glucose Cholesterol SARS-CoV-2
- NeoECG Holter Monitor Portable ECG Monitor AI-ECG Pocket ECG
- Patient Monitor Vital Signs Monitor End-tidal Capnography
- Fingertip Oximeter Handheld Oximeter Wrist Oximeter Wearable Oximeter All-in-One Health Monitor POCT Solution
- OBS Locking Compression Plate Locking Plates Conventional Plates Mini Plates System Screws Patella Ring - Patented Product Metal Pins Cable System
- Internal Fixation System Laminoplasty Plate System Percutaneous Kyphoplasty Fusion Cage Cervical Fixation System
- Hip System Knee System
- Suture Anchor Suture Button
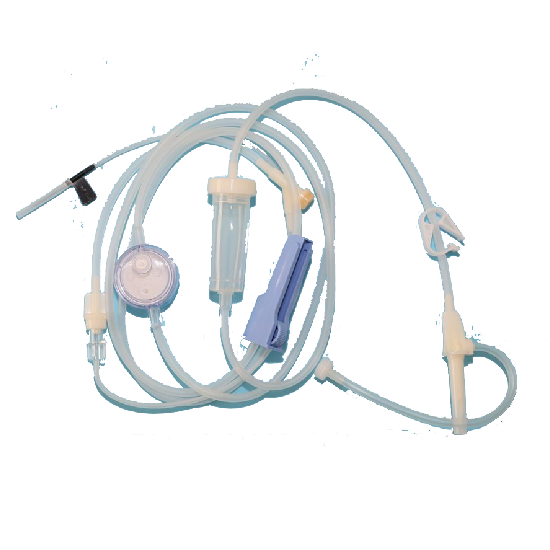
Certifications
NMPA
Safety Standards
GB/T 19001-2016 idt ISO 9001:2015
YY/T 0287-2017 idt ISO 13485:2016
Filter pore size
5μm/3μm/0.22μm
Features
1.This product does not contain DEHP, effectively avoiding the harm of DEHP:
Commonly used PVC infusion sets contain plasticizer DEHP, which accounts for about 30%-40%. DEHP can be precipitated and dissolved in the liquid during infusion and enter the human body. In particular, the infusion of alcohol-soluble, fat-soluble and anti-tumor drugs can accelerate its precipitation.
2.Low absorption to drug, improving the effectiveness of infusion:
The traditional PVC infusion set has been proved to have a high adsorption effect on some drugs, especially alcohols, lipids andanti-tumor infusion drugs. Its high absorption can lead to inaccurate medication, reduced e_x001d_cacy, prolonged hospitalization duration, and increased costs. The dripping pot and the inner layer of the tube are made of non-PVC materials, which have low absorption to the drug and can ensure the effectiveness of infusion.
3.Double-layer structure to ensure the safety of infusion:
The inner layer, in contact with the medical liquid, is made of TPU material to ensure the safety of the infusion; the outer layer is made of PVC material without plasticizer DEHP, which not only protects clinical nurses, but also facilitates the operations andmaintains the physical properties of the original infusion set without changing the operating habits of the medical personnel.
4.Advantages of medical-grade TPU:
The materials recommended by the US Food and Drug Administration (FDA), widely used in the clinical flied for nearly 60 years, safe and non-toxic; good biocompatibility; low adsorption of the drug to ensure the effectiveness; good elasticity and resistance to flexure, easy to handle; low friction coefficient on wet surfaces, difficult to produce air bubbles.
5.Precise filtration:
Imported nucleopore membrane, high filtration accuracy. Significantly reduce the incidence of infusion reactions (such as phlebitis, allergy, pyrogen-like reactions, etc.), and relieve the pain during infusion.
Thank You for Your Attention on Lepu Medical!
Email us with any questions or inquiries or use our contact data. We would be happy to answer your questions.
